Thermo Chemistry Test 1
10:18:00 Unknown 0 Comments
I am Mechatronics and Control Engineer from UET, Lahore. Helping Engineering students gives me pleasure.
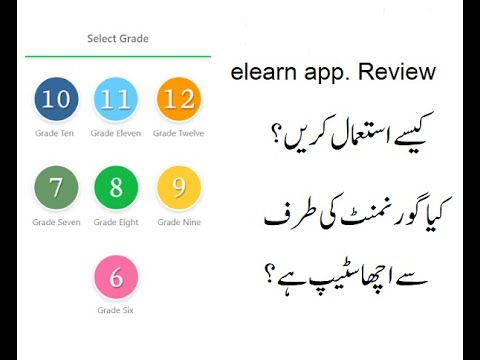
گورنمنٹ آف پنجاب کی طرف سےایک ایپ متعارف کروائی گئی ہے۔ جو کہ سٹوڈینٹس کے لئے کافی useful ثابت ہو سکتی ہے۔ اس ایپ میں کلاس 6 سے ...
0 comments:
Confused? Feel Free To Ask
Your feedback is always appreciated. We will try to reply to your queries as soon as time allows.